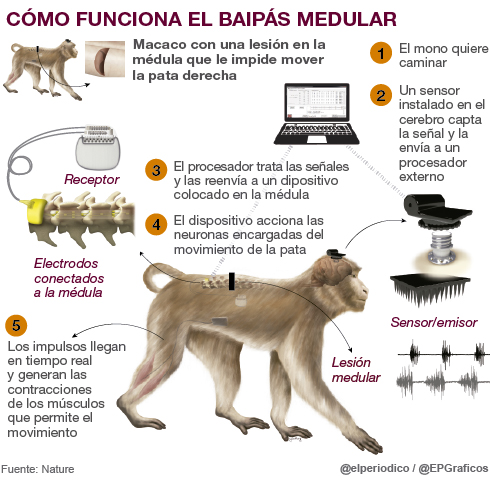
A device picks up signals from the brain and forwards them to a sensor that activates the endings responsible for controlling the muscles.
A macaque that could not move one of its hind legs because of a spinal cord injury, has managed to walk almost normally thanks to a “bypass” that wirelessly communicates the brain and the endings responsible for activating the muscles. This cerebrospinal neuroprosthesis, as defined by those responsible, has been developed at the École Polytechnique Fédérale de Lausanne, in Switzerland, in collaboration with other research centres and the company Medtronic.
The animal has a small sensor built into the brain that captures the emitted signals – from the macaque’s desire to move – and sends them to a processor. The data is treated and decoded and then forwarded to another device with electrodes that are placed in the lumbar area of the spinal cord, on the other side of the injury, and activate the neurons that direct the muscles of the affected leg. -“What we do is restore in real time and wirelessly the communication between the celebrity and the locomotor system“, says one of the authors in the project, Eduardo Martín Moraud, a Spanish engineer who works at the University of Oxford (Nuffiel College) and who had already been 5 years in the team of the Swiss School.
The details of the research, whose first satisfactory results were obtained in June 2015, have been published in the scientific journal ‘Nature’. One of the two monkeys in the experiment recovered the use of his paralyzed limb in the first week after the injury, without training, both on the treadmill and on the ground, while the second took about two weeks.
As reported by the EPFL, a clinical study has been launched at the University Hospital of Lausanne to validate the therapeutic effects of the technology in people with spinal cord injury, but only partially. For now, what is done is to check if the placement in the marrow of a device with electrodes, previously programmed, manages to restore the movement of the legs. It does not communicate with the brain.
THE PROCESS
As the researchers explain, when the brain decides to perform a movement or any other activity, there is “a transmission of electricity peaks between neurons” that can be measured and interpreted by a mathematical algorithm. In an intact nervous system, the signals that denote walking come from a small region of the brain called the motor cortex (or motor cortex).
Subsequently, the signals travel through the spinal cord, reach the neural networks located in the lumbar region and these activate the muscles of the legs to produce the movements.
Spinal cord injuries, partial or complete, prevent these signals from reaching neurons and cause paralysis. However, the motor cortex still maintains the brain activity that generated walking instructions. And the neural networks that activate the muscles in the paralyzed leg are also intact and can still generate leg movements.
On this occasion, “96 neural channels of the cortex that offer a lot of information” were measured, says Martín Moraud, who recalls that in previous experiments, for example, it had already been possible for human patients to activate a computer at a distance just by thinking about doing so. “We were interested in controlling only two events: when the leg is raised and when it is perched,” he continues.
Then, the electrical stimulation of a few volts, in a specific area of the spinal cord, modulates different networks of neurons that can activate specific muscles in the legs. “I have dealt specifically with the system that allows you to stimulate the marrow in real time,” continues Martín Moraud.
NO TRAINING REQUIRED
For the monkeys to regain mobility, “no physiotherapy or training was necessary,” says neuroscientist Erwan Bezard of the University of Bordeaux, who oversaw the experiments. All treatments have been carried out after control by the bioethics committees of the participating centers.
“For the first time I can imagine a completely paralyzed patient being able to move his legs through this brain-spinal interface,” neurosurgeon Jocelyne Bloch of the University Hospital of Lausanne (CHUV), who is responsible for placing the implants in the brain and spinal cord, said in a statement.
Source: The journal, Scientific Journal Nature.


